One of my recent bugaboos has been the increase in the number of papers claiming a “green” synthesis, reagent, process or solvent. It seems that I am not alone in my concerns of the misuse of words such as “green” and “sustainable” based on a recent paper called ‘What does it mean that “something is green”? The fundamentals of a Unified Green Theory’ (Green Chem., 2023, 25, 4625-4640). The paper starts with an excellent critique of the 12 Principles of Green Chemistry and shows how 6 of the 12 principles could be viewed as derivatives of (or secondary to) other principles, The 6 primary principles address criteria of waste, reagent toxicity, energy use, use of renewable materials, creation of degradable products, and user safety, whereas the secondary principles indicate the means to implement the primary principles. For example: Principle #2 of Atom Economy is really a subset of Principle #1 of Prevention (preventing waste is better than treating or cleaning up waste after it is created).
Before going any further I will quote the author, Pawel Nowek, at length because his sentiments about this article reflect closely my own increasing discomfort with the overuse of “green” as a catch-all term that has been imprecisely defined:
“It should be clearly emphasized that my goal is not to criticize the current sets of principles because their great merit for the propagation of ideas and stimulation of the development of new, more environmentally and human-friendly methods of synthesis and analysis is undeniable. The goal, however, is to indicate potential areas for improving the theoretical basis of Green Chemistry, and one of them is the possibility of formulating new, unified, and universal guidelines”.
The paper then goes into quite a long section that tries to define “green” and “greenness”. A few years ago I would have felt this section was academic semantics, but later in my career managing scientific teams and projects I came to the conclusion that root cause of most arguments were fundamentally due to a lack of definition or alignment about what was being discussed, so I encourage readers to work through this section and think about areas in your own research where a more precise definition of the problem, goals or metrics for success might be beneficial. Ultimately this section of the paper concludes that any measure of “green” or “greenness” cannot be absolute. Rather, one can only indicate which object (product, process or method) appears greener according to a specified evaluation model. This should lead to a discussion based on facts supported by transparently processed data, rather than opinions or wishes portrayed as facts, which ultimately will allow the field of Green Chemistry to advance more rapidly.
In the middle of the paper a new concept of “White Chemistry” is introduced where the concept of “white” is at the intersection of of “Green”, “Red” and “Blue” chemistry as indicated in the figure below. To me this much better reflects the reality of working as an industrial process chemist where “Red” indicates the effectiveness of the process being developed to deliver the target molecule in sufficient quantity on-specification, and “Blue” represents the need to deliver on-time and on-budget. Without being able to balance all of these attributes in a fit-for-purpose manner the societal benefits that we are trying to deliver cannot be met. The author concludes this section of the paper by pointing out that we can only really assess greenness and sustainability once a certain level of utility has been met which I wholeheartedly agree with, especially where academic groups are comparing chemistry that they may have implemented on a few grams and are typically analyzed by crude generic methods, rather than the detailed specific methods developed to ensure patient safety. Without such analysis and implementation on multi kilo or multi ton scale, is the academic method even able to deliver a fit-for-purpose product?
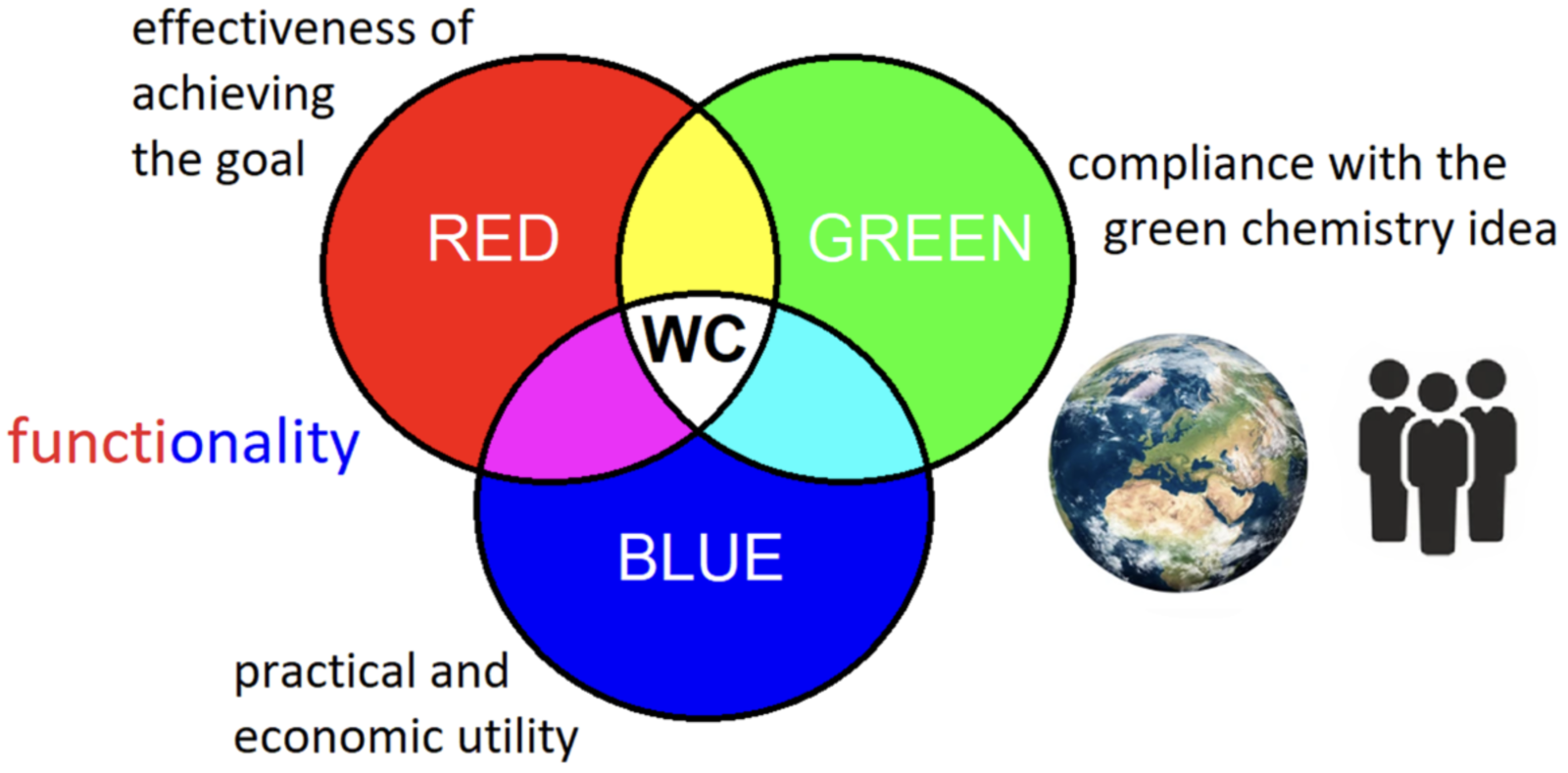
The final third of the paper works toward a new unified green theory. Five elements that have to be considered in any assessment are identified:
- The reduction of the emission of greenhouse gases
- The consequences associated with the use of chemical reagents that are dangerous to the environment and/or human health
- The depletion of the Earth’s natural resources and possibility of recycling
- Possible physical impacts, such as injuries caused by temperature, radiation, high pressure, high voltage
- The consequences of exposure to biological hazards, both to the operators in the labs and manufacturing facilities, as well as the wider societal risk should one of these lead to an epidemic
There is then a discussion about how these elements might be defined mathematically as functions of the severity of the hazard, the quantity of the hazard, and the effectiveness of methods to mitigate that hazard. For example the quantity of greenhouse gases emitted will depend on the scale of the process but also where the process is implemented: some areas of the world already have electricity primarily derived from renewable sources, such as hydroelectric, solar and wind, whereas others are still reliant of coal or oil.
Finally five unified principles of greenness are proposed in a hierarchical order:
- Limitation. In order to strive for greenness, limit all destructive impacts on the environment and humans caused by your actions directly or indirectly.
- Cooperation. Three types of actions lead to greenness: elimination of hazards, reduction of the quantity of hazardous factors, and increase in the effectiveness of counteracting (prevention). It is important to merge efforts and act simultaneously at all these levels.
- Compromise. Do not strive to achieve your goals at any cost. Search for the optimal compromise in a specific situation and circumstances guided by common sense.
- Verification. Whenever possible, rely on hard empirical data, avoid conjectures and subjective assumptions. Use models with care. Be critical of yourself and your beliefs.
- Clarity. Use strict and transparent language. Define the terms you use and use the terminology consistently.
Beyond Principle #1 (Limitation) which is more specific to Green Chemistry I would argue that any well run scientific project would benefit from applying Principles #2 – #5!
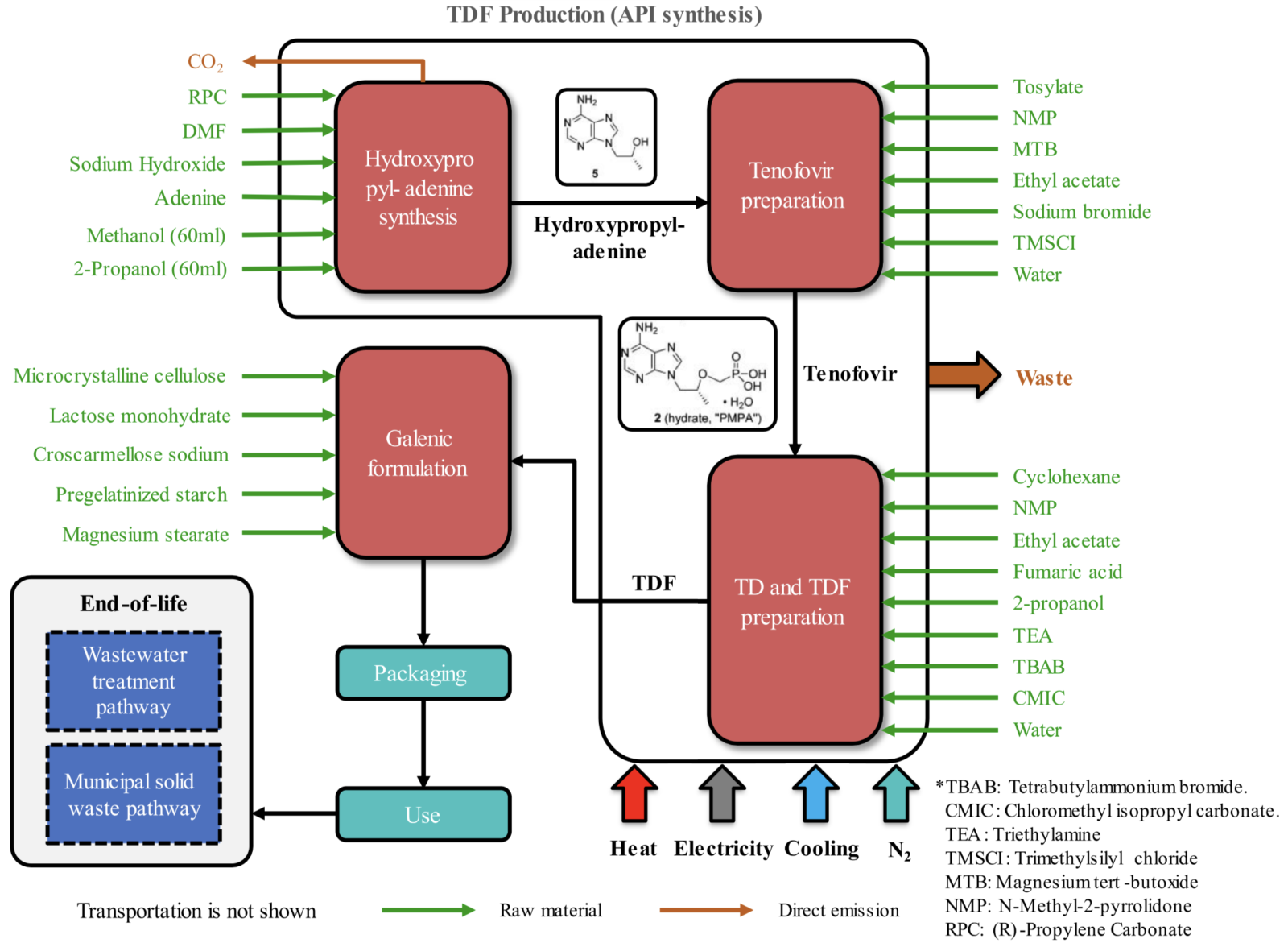
A recent full lifecycle analysis for the global production of the antiretroviral therapy tenofovir disoproxil fumarate (ACS Sustainable Chem. Eng. 2023, 11, 6510−6522) illustrates just how comprehensive a set of data are needed to complete an accurate assessment of the greenness of a process, as shown in the figure below.
Another recent paper (Org. Process Res. Dev. 2023, 27, 822−830) describes the most thorough analysis of the environmental impact of of water performed in water using designer surfactants by taking into account the intrinsic carbon footprint of not only the reaction mixture but also the impact of synthesizing the surfactant, the work-up (including the possibility of recycling both solvent and precious metals), and disposing of the wastewater.
Both of these analyses are likely beyond the scope that an R&D chemist can reasonably expected to bear in mind while developing a new synthesis route or chemical process, but both papers identify a few timeless common themes that we teach in a number of Scientific Update courses, including the foundational Chemical Development course
- A holistic view of process development needs to be taken to balance all competing factors
- Reactions should be run as concentrated as possible
- Solvents should be recycled if at all possible, which requires developing a solvent strategy across many process or synthesis steps
- Clearly problematic solvents, such as DCM, DMF, NMP and DMAc, should be replaced whenever possible
- Good metrics and understanding leads to better processes in the long term
Ultimately, good chemistry is green chemistry and we should all be constantly striving to raise the bar and lower the impact of our work on the environment while delivering valuable products to society.








