Esomeprazole is a proton pump inhibitor developed by Astra Zeneca in 2001 and marked under the trade name Nexium. Esomeprazole is the S-enantiomer of omeprazole (the racemate) and is an example of the controversial so-called “chiral switch” in with single isomers are developed and marked as having improved pharmacodynamic or phmacokinetic properties over the originally marketed racemate and the patent life of the compound is extended, the so called “evergreen strategy”.[1] Sales of Nexium in 2006 peaked at 5 billion dollars.
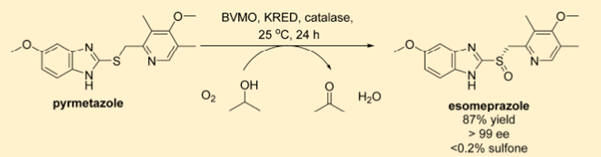
Esomeprazole contains a somewhat unusual sulfoxide-containing API and is manufactured using a titanium-based oxidation process based on chemistry developed by Kagan and Sharpless.[2] The chemical problems inherent in this approach, including water sensitivity, over-oxidation to sulfone, together with the engineering challenges relating to process safety and environmental impact highlighted a need for an improved asymmetric synthesis. A recent paper by David Entwistle and his team at Codexis describe the development of an enzymatic oxidation of pyrmetazole using an engineered Bayer-Villager monooxygenase (J. Org. Chem. 2018, 83, 7453).
The team used BVMO in combination with an NADPH co-factor recycling system (ketoreductase-mediated propan-2-ol oxidation). Initial low efficiently with wildtype BVMO was rapidly improved using Codexis’ CodeEvolver directed evolution platform. After 19 evolutions a 105 fold improvement was obtained, with >99% ee and 0.1% sulfone impurity. Formation of hydrogen peroxide during the reaction, which can lead to enzyme deactivation and an achiral sulfur oxidation, was prevented by the addition of catalase. Mass transfer challenges were encountered on scale-up, however experimental for a successful 30g lab demonstration batch are reported in the paper.
[1] Australian prescriber 2004 (DOI: 10.18773/austprescr.2004.039)- Inside the isomers- the tale of chiral switches
[2] Tett. Lett. 1984, 25, 1049; Tett. Asym. 2000, 11, 3819








