Multipurpose facilities for batch processing remain ubiquitous in the process industries for relatively short campaigns of diverse products. Whether the products be active pharmaceutical ingredients, foods, agrochemicals, or other fine and specialty chemicals, those producing them face a shared problem: how to satisfactorily remove residues of the previous campaign from the plant before commencing on the next to prevent adverse impacts on quality. This concern is most acutely felt in pharmaceutical products where cross-contamination potentially has clear implications for patient safety.
The time and resources expended on plant changeover and cleaning throughout the process industries are substantial, and yet planning for this activity is often the Cinderella of process development. Someone working for one of the major pharmaceutical companies told me that, “Changeovers are our biggest product”, going on to explain that changeovers represented about one third of available plant operating time. Similar during a study at a CMO in the UK, it was reported to me that over a single year, actual changeover time was 165% of the time planned. This was across many changeovers of which 30% were new product introductions. Both of these snapshots are indicative of inefficient cleaning procedures. Neither goes into the details of additional solvents or energy consumed in ineffective cleaning but anyone familiar with methods typically employed during plant cleaning can easily start to form their own picture.
How much do we know about the mechanisms in cleaning that remove deposits from a surface? The scientific literature appears to be sparse, although there is some active research in the field, for example at the Universities of Limerick[1] and Birmingham[2], and at the Institute of Chemical and Engineering Sciences in Singapore[3]. In broad terms for the pharmaceutical and fine chemicals sectors however, things do not appear to have advanced much since1959 when Herbert Sinner at Henkel proposed his cleaning circle for the four driving forces for cleaning; chemical, mechanical, temperature and time.[4]
Many organisations’ procedures for planning cleaning focus on finding a solvent for the main product through solubility studies. This only addresses part of the problem. What about the other raw material and impurity residues? Also, solubility is a thermodynamic property but, cleaning depends on the kinetics of dissolution. Typically, little emphasis is placed on understanding the mechanical aspects of cleaning, but (because they will influence the kinetics) they are key to the effectiveness of any cleaning operation.
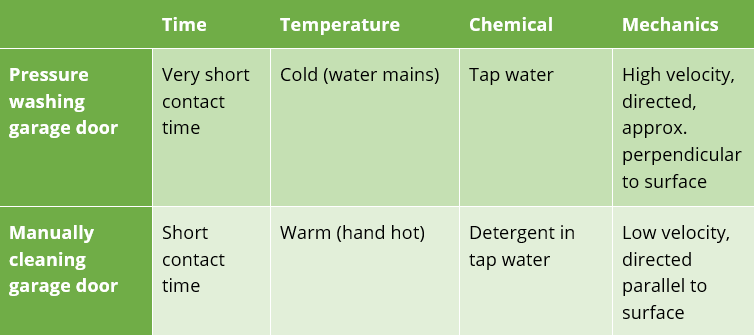
I encountered a simple example of this, when cleaning my garage door for repainting. I tried a pressure washer first, which I expected to be very quick and efficient. This however completely failed to remove a grimy biofilm which had built up across the whole door. Cleaning by hand with a sponge and warm, soapy water did a much better job. Comparing the two cleaning methods using Sinner’s four driving forces is informative.

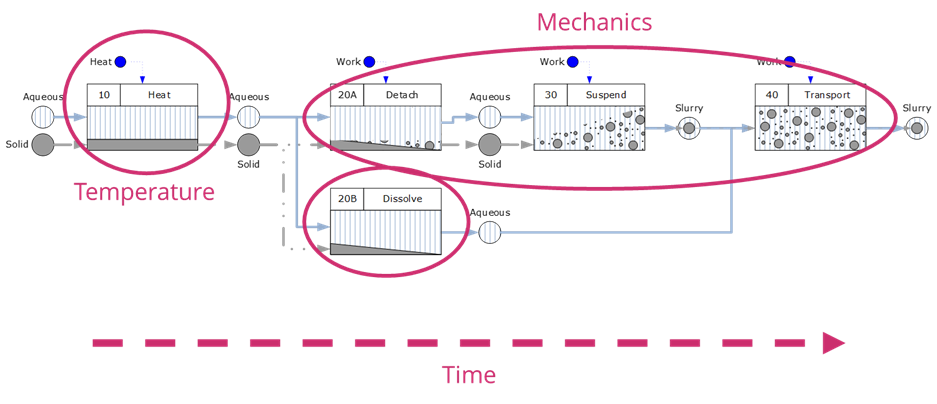
This kind of thinking can be readily developed and applied to cleaning process equipment. The diagram below considers the processing experience of the residues during cleaning with the four driving forces superimposed. Once again the importance of mechanics is emphasised.
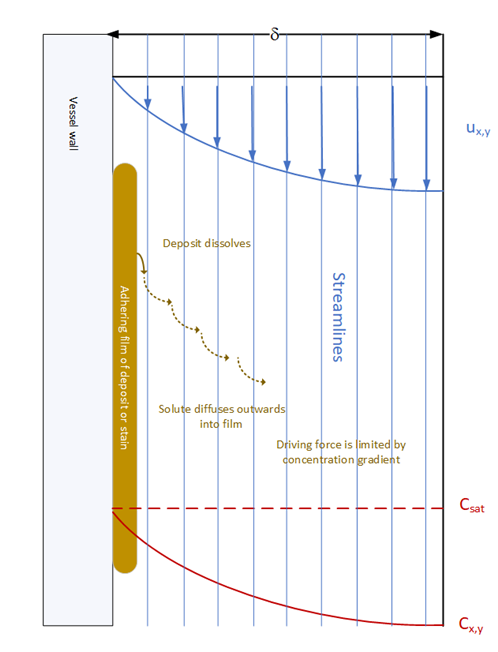
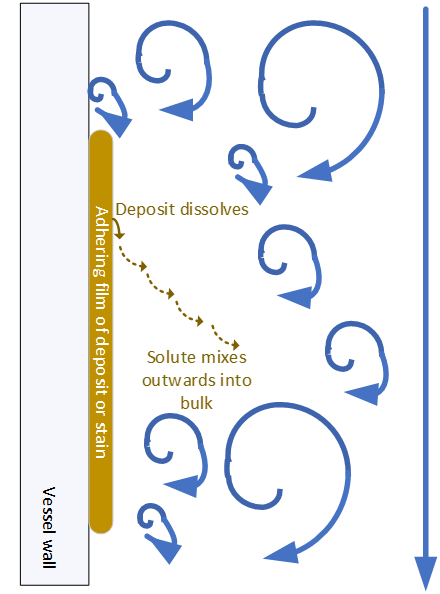
Considering as an example, cleaning a vessel wall with a spray ball, most of the energy of the spray is dissipated in flight. A film of liquid develops and runs down the wall. Unless very large amounts of liquid are sprayed, the flow regime of the falling film is laminar. This adversely impacts on dissolution because solute is saturated at the surface can only move away from the residue surface by the slow process of molecular diffusion. The shearing effect of the falling film will dislodge gross deposits and large particles. The analysis suggests that a spray ball will be effective for initial cleaning but much less effective with firmly attached residues. Filling the vessel with solvent and agitating, leads to a very different, turbulent flow regime adjacent to the wall which will improve the rate of dissolution.
As pressure increases to move to more sustainable process industries, there is a need to take a wider view and consider the whole processing system. This is evidenced by the attention being called to the energy consumption of the HVAC and extraction systems in the pharmaceutical sector. Focus will soon turn to other ancillary activities, such as cleaning. Developing an improved process understanding provide an opportunity to systematically develop robust cleaning procedures, saving unproductive time, and reducing costs, solvent wastes and energy usage helping to make the production system more sustainable.
About the Author:
Rob Peeling obtained a BSc in Chemical Engineering from UMIST in 1983. This included two industrial placements in the chemical laboratories at Monsanto and at Shell Carrington. Since then he has worked for Tioxide , ICI Katalco and Johnson Matthey’s Technology Centre before joining his current company Britest in 2016. Rob is involved with teaching on a number of training courses and during 2022, he is delivering a webinar explaining more fully how better understanding can underpin improved cleaning procedure development to benefit efficiency and sustainability ‘Plant Changeover and Cleaning – Better Understanding for Better Results‘, February 10th, 2.00 pm (UK). Later in the year a NEW Online course will be available looking in detail at the fundamental principles underlying successful cleaning and a methodology for developing robust cleaning protocols: ‘Understanding Process Plant Cleaning for More Efficient Changeovers‘, June 8-10, 2022.
[1] Zhang, C, et al., Rethinking Cleaning Validation for API Manufacturing, Pharmaceutical Technology, September 2018, pp 42-54
[2] https://www.formulation.org.uk/images/stories/FormulaX/Presentations/Formula%20X%20-%20C-22%20-%20Perrakis%20Bistis.pdf (checked June 2020)
[3] https://www.a-star.edu.sg/pips (checked June 2020)
[4] Sinner, H, Über das Waschen mit Haushaltwaschmaschinen: in welchem Umfange erleichtern Haushaltwaschmaschinen und -geräte das Wäschehaben im Haushalt?, Volume 8 of Haus und Heim, Ed. 2. Haus+Heim-Verlag, (1960)








